Ultrasonography of the gastrointestinal tract
Ultrasonography of the gastrointestinal tract in dogs and cats
Ultrasonography of the gastrointestinal (GI) tract should be included in every routine abdominal ultrasound examination. Specific indications for gastrointestinal ultrasonography include:
- Chronic vomiting and diarrhoea
- Palpable abdominal masses or thickening of gastrointestinal structures
- Abdominal pain
- Weight loss and/or anorexia
- Suspicion of ingested foreign bodies
- Investigating possible hernias
- Staging neoplasia
Ultrasonography of the GI tract can provide useful information including the overall wall thickness, relative thickness and echogenicity of the wall layers, motility, contents, and the appearance of associated structures such as the mesentery and mesenteric lymph nodes. Knowledge of the normal and abnormal appearances of the GI tract and the ability to interpret artefactual shadowing can reduce the requirement for multiple imaging modalities, and in selected cases, such as obstructive foreign bodies or intestinal perforation, provide a singular sensitive and specific diagnostic test.1,2
Examination Technique
One of the major limiting factors when attempting to image the GI tract is the presence of gas within the lumen. Reverberation artefact caused by gas prevents visualisation of the far field, often preventing complete evaluation of structures. Preparing the patient by withholding food for 12 to 24 hours prior to examination is recommended. It is important to remember that certain drugs and the stress or confinement of hospitalisation can also alter GI motility. A high frequency linear or micro-convex transducer (7mHz or higher) should be used to enable assessment of wall layering. The smaller footprint of a micro-convex probe facilitates the use of the intercostal spaces to achieve views of GI structures.
Dorsal, left or right lateral recumbency can be utilised and all confer different advantages. Dorsal recumbency is likely to allow complete visualisation of the gastrointestinal tract, whilst lateral recumbency (left or right) will assist with redistribution of luminal gas or fluid, thus allowing more compete visualisation of individual sections of the GI tract (Figure 1). In deep chested dogs, an intercostal right lateral view may be required to visualise the proximal duodenum and/or gastric pylorus.

Figure 1. This ventrodorsal abdominal radiograph has been annotated to give a schematic representation of the position and course of parts of the GI tract in a dog. In cats, the stomach is orientated more obliquely, and the pylorus and duodenum are positioned closer to the midline.
Normal Appearance
All sections of the GI tract share a common layered ultrasonographic appearance (Figure 2). However, the overall and relative thickness of parts of the GI tract layers varies with different sections of the GI tract.

Figure 2. A Jejunal loop imaged in longitudinal section – Wall layering of the gastrointestinal tract can be divided in to 5 distinct layers:
- Lumen – sometimes referred to as the luminal-mucosal interface. The lumen’s appearance can be variable depending on luminal contents, in this image the lumen is empty and exhibits a mucous pattern (hyperechoic line).
- Mucosa – hypoechoic in appearance, the width is variable dependent on the section of gastrointestinal tract.
- Submucosa – throughout much of the gastrointestinal tract this appears relatively narrow and hyperechoic.
- Muscularis – hypoechoic in appearance with variable thickness.
- Serosa – a thin hyperechoic layer, clear identification is often difficult.
Gastrointestinal wall thickness is measured from the inner edge of the mucosal layer outwards to the outer edge of the muscularis layer (Figure 3).

Figure 3. An ultrasound image showing 2 sections of small intestine. Two callipers (crosses with linked dotted line) have been placed to measure intestinal wall thickness, from the inner edge of the mucosa outwards to the outer edge of the muscularis layer.

The table above provides the reference ranges of normal wall thickness of different sections of the GI tract in dogs and cats (some breed variation may occur in dogs). Changes in the values for wall thickness should always be considered in the context of the clinical presentation along with the distribution of changes and alterations in the pattern of wall layering.
Stomach
The stomach has a variable appearance, depending upon luminal content (Figure 4). Intraluminal gas or ingesta can obscure the far wall whilst fluid content can highlight the walls, allowing the layers to be observed more clearly. A collapsed, empty stomach can make wall thickness difficult to evaluate because the lumen may be indistinguishable from the walls. Rugal folds are clearly visible in an empty stomach and are more commonly observed in the fundic rather than pyloric region. When measuring wall thickness, an inter-rugal fold measurement should be taken (i.e. between rugal folds). Observing peristaltic contractions can be used to assess motility; these average at around five contractions per minute.3

Figure 4. Stomach Wall – The appearance of the stomach is variable depending on luminal contents. Collapse of the stomach walls in the image on the left prevents easy identification of the stomach lumen. Artefact caused by the ingesta and gas in the image on the right prevents visualisation of the far wall of the stomach.
Duodenum
The canine duodenum is located running in a cranio-caudal direction along the right lateral body wall, following a superficial and more linear path than the jejunum (Figure 5). In contrast, the feline duodenum lies more ventrally, towards the midline. In both species the duodenum can be differentiated from jejunum by anatomical location and slightly wider wall thickness. 4,5

Figure 5. Duodenum – In this sagittal plane image of the right dorsal abdomen of a dog, the duodenum (arrow) an be visualised in longitudinal section. Deep to the duodenum, multiple loops of jejunum in transverse section can be seen.
Jejunum
Jejunal loops can be visualised throughout the abdomen. Motility ranges from 1-3 peristaltic contractions per minute. 3
Ileum
The ileum is most readily identified using a right lateral approach. The prominent submucosa and contracted state give the lumen a star shape or rosetted, sphincter-like appearance (Figure 6). Although commonly found in cats, the ileo-caeco-colic junction is more rarely visualised in dogs, likely due to the presence of gas in the colon or caecum.5

Figure 6. Ileum – The ileum and a loop of jejunum are imaged in cross section in the same image. Note the difference in the appearance of the lumens; a rosette or star shaped appearance in the ileum, compared to a more linear appearance in the jejunum. The submucosa has a more pronounced and hyperechoic appearance in the ileum.
Colon
In comparison to the more proximal sections of GI tract, the colon has a much thinner total wall thickness, with layering which is more difficult to evaluate without a high frequency probe (Figure 7). Intraluminal gas and faecal contents commonly cause a distal acoustic shadowing artefact, which obscures the far wall.

Figure 7. Large Intestinal Wall Thickness – In this sagittal plane image of the right abdomen of a dog the ileo-caeco-colic junction (ICCJ) can be seen. The duodenum is visible in the near field (SI); note the marked difference in overall wall thickness between the small (SI; both walls highlighted) and large (LI; one wall highlighted) intestine.
Ultrasonographic appearance of gastrointestinal pathology
When performing abdominal ultrasound of the GI tract, the following factors should be evaluated:
- Total wall thickness or changes in relative wall layer thickness (Figure 8, 9)
- Alteration in the relative echogenicity of the layers, particularly subtle changes to the mucosa
- Alteration in the pattern or distinction of wall layering (Figure 10)
- Subjective gastric or intestinal distension
- Intestinal plication or corrugation (Figure 11)
- Alteration in motility: hypermotility, spasmodic motility or intestinal ileus
- Identification of luminal contents (Figure 12)
Assessment of associated anatomy including lymphadenopathy, echogenic changes to the surrounding mesentery and presence of abdominal free fluid should also be performed.1,2,5

Figure 8. Muscularis Layer Thickening – In this image of a cat, sections of jejunum can be seen. Although not thickened overall (one wall thickness is being measured by the yellow callipers – 0.27cm), there is a relative increase in the thickness of the hypoechoic muscularis layer. This change is associated with lymphoma and inflammatory bowel disease in cats. 8
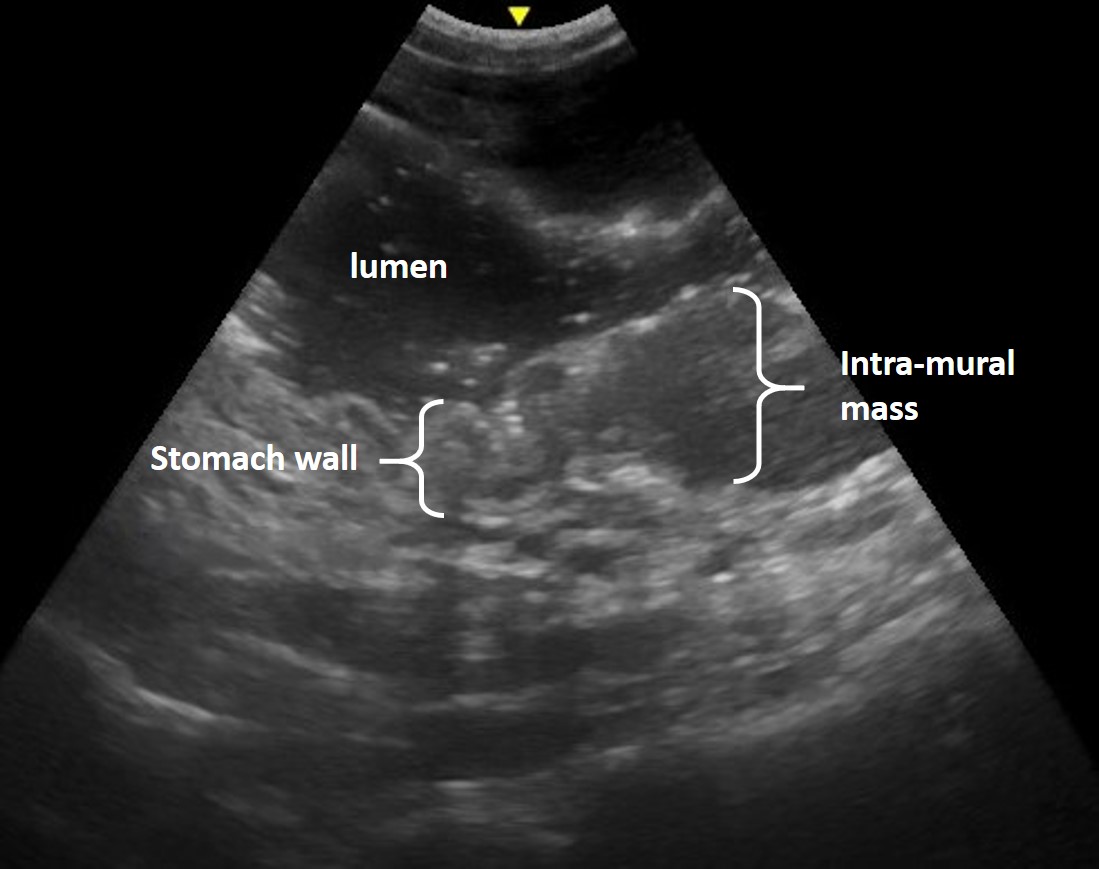
Figure 9. Feline gastric lymphoma – The fluid-filled lumen of the stomach highlights the normal, layered appearance of the stomach wall in the left of the image. There is an intramural mass with associated loss of layering, thickened stomach wall and relative change in echogenicity to the right of the image.

Figure 10. Intussusception – This image shows a transverse plane image of an intussusception. Here, a multi-layered ‘target’ or ‘bullseye’ pattern can be seen, caused by one section of small intestine invaginating within itself.

Figure 11. Corrugation – This image shows a section of jejunum (arrow). The intestinal wall has an undulating pattern typical of corrugation. This finding has been described in a number of pathologies including enteritis, peritonitis, pancreatitis and neoplasia. 9 Plication, which is a change associated with foreign bodies particularly linear foreign bodies, appears as a more eccentric arrangement of the intestine compared with the more regular undulation seen in
corrugation.

Figure 12. Gastric Foreign Body – In this sagittal oblique image of the cranial abdomen of a dog, a plastic foreign body in present within the gastric lumen (arrow). The dense plastic of the foreign material is preventing the ultrasound waves penetrating deeper into the patient and creating an acoustic shadow artefact (*).
Sampling Considerations
Ultrasound-guided fine needle aspiration of GI wall lesions can be performed using a 22 – gauge needle. 10 It is important to ensure that the needle does not enter the lumen of the GI tract during the procedure. However, obtaining samples from some areas of the GI tract can be difficult, as can differentiating between inflammatory and neoplastic conditions. For these reasons, an endoscopic or full thickness surgical biopsy is a more reliable diagnostic test.
References
- Tyrell D. Beck C. (2006) Survey of the use of radiography vs. ultrasonography in the investigation of gastrointestinal foreign bodies in small animals. Veterinary Radiology & Ultrasound 47: 404–408.
- Boysen S.R. Tidwell A.S., Penninck D.G. (2003) Ultrasonographic findings in dogs and cats with gastrointestinal perforation. Veterinary Radiology & Ultrasound 44: 556-564.
- Penninck D.G. Nyland T.G., Fisher P.E., Kerr L.Y. (1989) Ultrasonography of the normal canine gastrointestinal tract. Veterinary Radiology 30: 272-276.
- Newell S.M., Graham J.P., Roberts G.D., Ginn P.E., Harrison J.M. (1999) Sonography of the normal feline gastrointestinal tract. Veterinary Radiology & Ultrasound 40: 40-43.
- Larson M.M and Biller D.S. (2009) Ultrasound of the Gastrointestinal Tract. Veterinary Clinics of North America: Small Animal Practice 39: 747-759.
- Gladwin N.E., Penninck D.G., Webster C.R.L. (2014) Ultrasonographic evaluation of the thickness of the wall layers in the intestinal tract of dogs. American Journal of Veterinary Research 75: 349-353.
- DiDonato P., Penninck D., Pietra M., Cipone M., Diana A. (2014) Ultrasonographic measurement of the relative thickness of intestinal wall layers in clinically healthy cats. Journal of Feline Medicine and Surgery 16: 333-339.
- Daniaux L.A., Laurenson M.P., Marks S.L., Moor, P.F., Taylo S.L., Chen R.X., Zwingenberger A.L. (2013). Ultrasonographic thickening of the muscularis propria in feline small intestinal small cell T-cell lymphoma and inflammatory bowel disease. Journal of Feline Medicine and Surgery 16: 89–98.
- Moon M.L., Biller D.S., Armbrust L.J. (2003). Ultrasonographic Appearance and Etiology of Corrugated Small Intestine. Veterinary Radiology & Ultrasound 44: 199–203.
- Penninck D.G., Crystal M.A., Matz M.E., Pearson S.H. (1993). The technique of percutaneous ultrasound guided fine-needle aspiration biopsy and automated microcore biopsy in small animal gastrointestinal diseases. Veterinary Radiology & Ultrasound 34: 433–436.
Related Learning


