Screening for OPA in sheep using transthoracic ultrasound
Ultrasound equipment carried for routine repro work (equine or bovine) can have many other applications; ovine transthoracic ultrasonography being one of them.
It is likely that veterinarians practicing farm animal medicine these days will carry an ultrasound scanner with a linear array transducer . Also it is also likely that most farm animal veterinarians will examine a number of sheep for varying reasons during their daily visits. Ultrasound equipment carried for routine repro work (equine or bovine). Can have many other applications; ovine transthoracic ultrasonography being one of them.
Ovine pulmonary adenocarcinoma (OPA) is one of a number of respiratory diseases prevalent in the UK sheep population. It is a disease that can cause significant annual losses to flocks; sometimes 4-5%, but more commonly 1-2% per year.
OPA (or jaagsiekte) is a contagious viral respiratory disease of sheep. Caused by the jaagsiekte sheep retrovirus, that results in the development of pulmonary tumours in some animals. Morbidity and mortality rates can be high, especially in newly infected flocks. Losses are generally fewer where the retrovirus has been present for longer.
OPA mostly affects sheep over 2 years of age. With the highest incidences occurring in those between 3 and 4 years old, and it is rarely seen in sheep less than 9 months old.The morbidity rate in a flock is often not as high as the incidence of infection as clinical signs may not develop during the productive life span.
Transmission of virus is via aerosol or direct contact with infected respiratory secretions. Clinical signs can take several years to develop due to the potentially very long incubation period. When they do develop, signs are vague (progressive respiratory disease that resembles pneumonia) until the disease becomes severe. There is currently no treatment for the disease. Therefore early detection and isolation from the flock are imperative for control of the disease within the flock.
Work at the Moredun Research Institute in collaboration with Capital Veterinary Services has shown that transthoracic diagnostic ultrasound. (using a Mindray DP50 scanner with curvilinear transducer but also possible with many other scanners, e.g. Easi-Scan with linear transrectal transducer). Can reliably identify even small tumours (1-2cm and larger) and allows veterinarians to identify OPA at stages before clinical signs are evident (reducing the spread of the virus) and while sheep still have cull value. The specificity and sensitivity of ultrasound scanning for OPA is generally very good. (Cousens & Scott, 2015), however false positives can occur. And it must be remembered that scanning cannot guarantee a sheep to be free from disease as the very smallest tumours will go undetected.
The technique for ovine transthoracic ultrasound using equipment of the above specification is quick and simple. A small area of skin (approx. 5cm wide) extending vertically from the point of the elbow is used and ultrasound gel applied liberally. The transducer is placed within the 5th to 7th intercostal spaces and the thorax evaluated (fig. 1), usually from dorsal to ventral to increase the likelihood of visualising normal lung tissue first as OPA usually affects the ventral lung.
Initially, set the frequency to around 7.5MHz and adjust the scanning depth to 6-8cm, enabling the evaluation of pleurae and superficial lung parenchyma. Increase the depth as necessary, typically up to 10-12cm, and correspondingly reduce the frequency to assess the full extent of any pathology found.
Normal aerated lung can be distinguished by a hyperechoic, white line close to the transducer and the associated reverberation artefact (fig. 2). Areas of consolidation and pathology can be identified as a loss of the bright white line and appear as hypoechoic, often well-demarcated areas compared to the surrounding tissue (fig. 3 and fig. 4). Lesions will often be found ventrally in the 5th and 6th intercostal spaces.
All in all, a quick and easy technique that could help to save many farms from sizeable losses related to OPA, but can also differentiate other lung pathologies (e.g. pulmonary abscess) that may require alternative treatment.
You can find additional information about OPA and the ongoing efforts to identify new diagnostic opportunities on the Moredun Research Institute website. Alternatively, you can contact Dr. Chris Cousens for more details.
References
Scott, P. (2016) Practical use of ultrasound scan in small ruminant medicine and surgery. Vet Clin North Am Food Anim Pract, 32(1): 181-205.
Griffiths, D. J., Martineau, H., Cousens, C. (2010) Pathology and pathogenesis of ovine pulmonary adenocarcinoma. The journal of comparative pathology,142(4): 260-283.
Griffiths, D., Cousens, C., (2012) Ovine Pulmonary Adenocarcinoma (OPA or Jaagsiekte). Moredun foundation newssheet, vol. 5 No. 16.
Cousens, C., Scott, P. R., (2015) Assessment of transthoracic ultrasound diagnosis of ovine pulmonary adenocarcinoma in adult sheep. Veterinary Record, 177(14):366.
Zhang, K., Kong, H., Liu, Y., Shang, Y., Wu, B., Liu, X. (2014) Diagnosis and phylogenetic analysis of ovine pulmonary adenocarcinoma in China. Virus Genes, 48(1): 64-73.

Fig. 1
The imager extends and holds the forelimb, exposing the 5th to 7th intercostal spaces.
Image courtesy of Dr. Chris Cousens
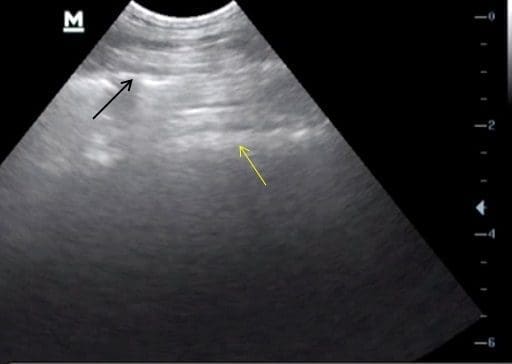
Fig. 2
The hyperechoic white line (indicated by the black arrow) deep to the transducer and the reverberation artifact (highlighted by the yellow arrow) distinguish normal lung. Note the depth setting on the right hand side is 6cm and the focal point set appropriately.
Image courtesy of Dr. Phil Scott
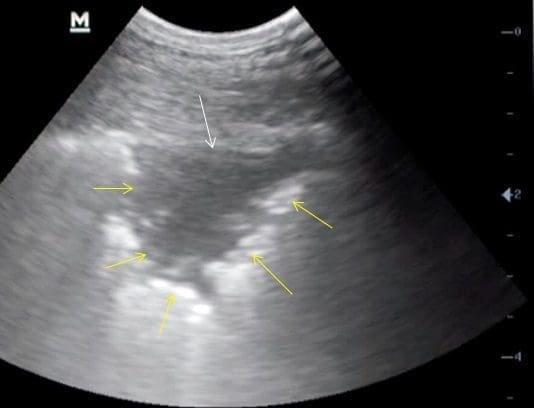
Fig. 3
A small (1-2cm) pulmonary lesion, with loss of the hyperechoic white line (white arrow) and a well-demarcated area of pathology associated with OPA (yellow arrows) in this case. Note the depth setting here is merely 4cm.
Image courtesy of Dr. Phil Scott
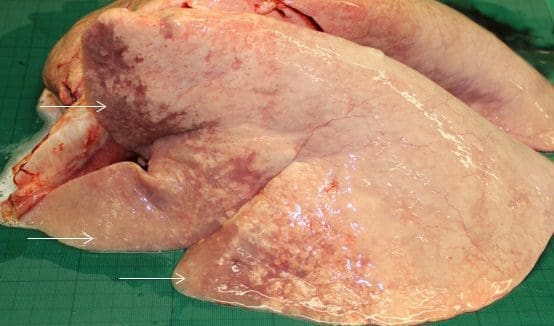
Fig. 4
Post mortem image (left lung) demonstrating the common ventral distribution of OPA lesions (arrows).
Image courtesy of Dr. Phil Scott